Introduction
The gas storage mechanism in a coalbed methane reservoir is different from the mechanism in a conventional gas reservoir.
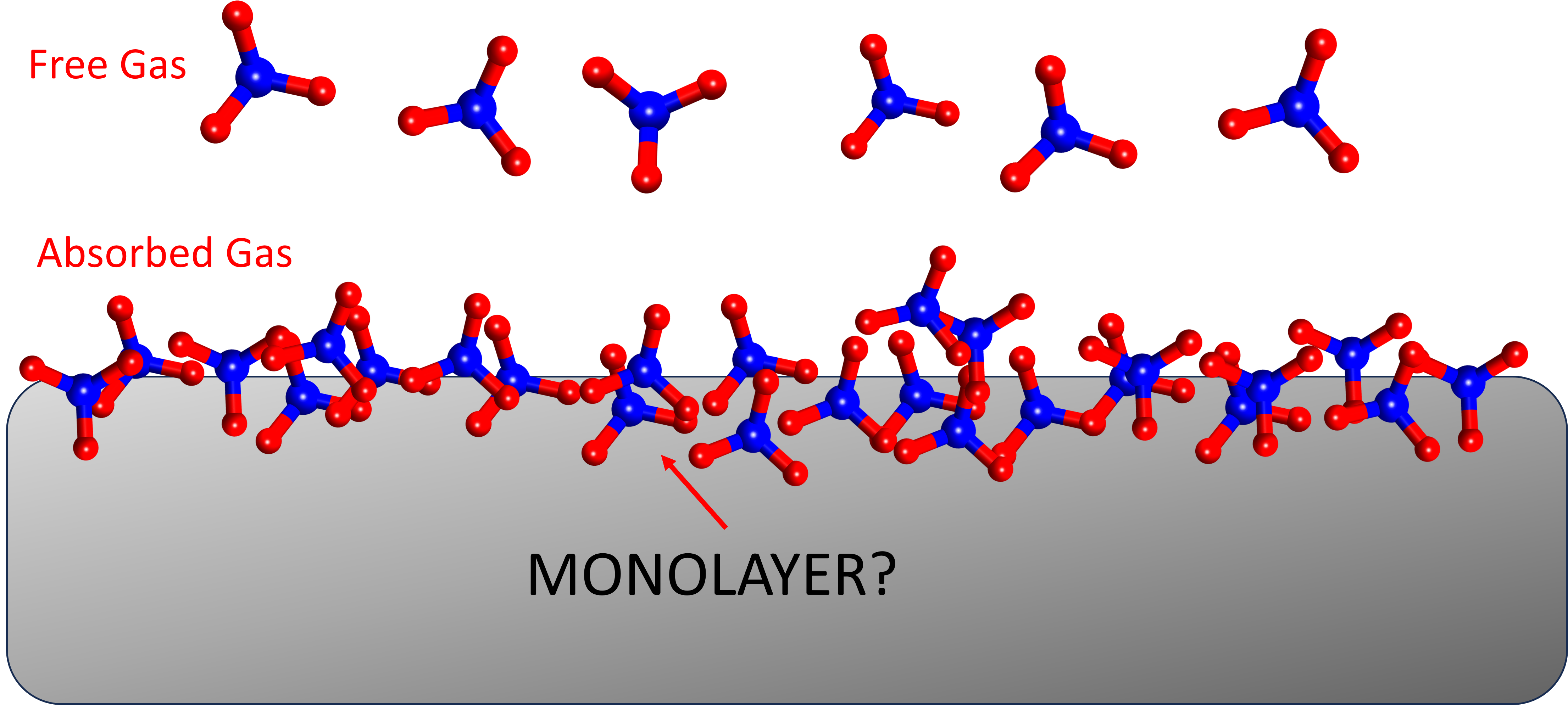
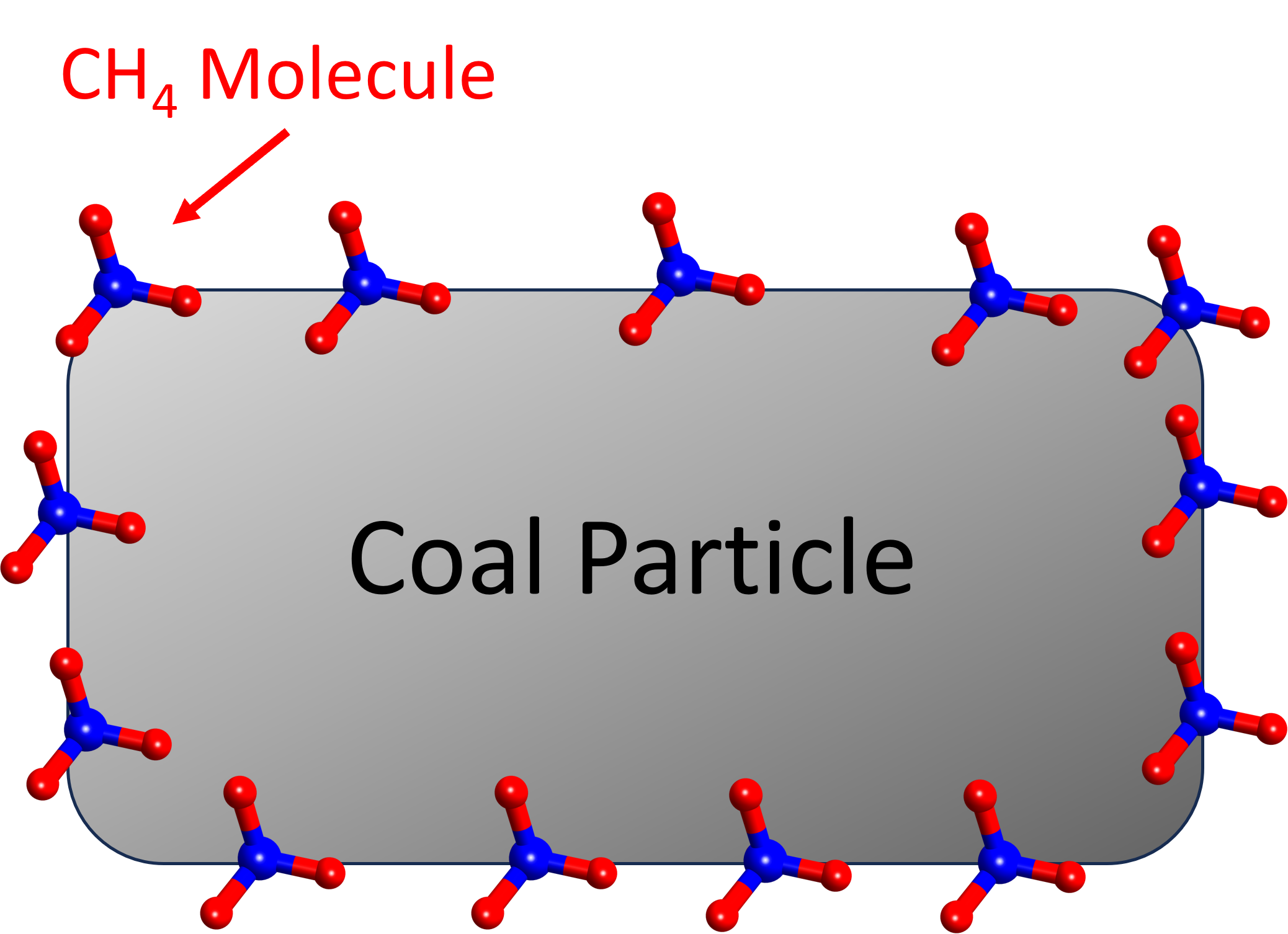
The methane gas is formed during the coalification process; that is, the coal formation from plant material conversion. During the coalification process, methane occurs as a by product and is adsorbed into the internal surface of the coal micropore system (as shown below). Therefore, a coalbed methane reservoir is also considered as both source rock and reservoir rock. In the images below, methane is shown as a monolayer on a coal surface.
Langmuir Isotherm
Coal has the ability to store a large amount of gas. Methane (and/or other combinations of gas) is adsorbed in the coal bed methane under the adsorption mechanism. In the simple scenario, gas is adsorbed in the internal surface of coal matrix. The adsorbed methane on the internal surface of coal matrix is modelled a common model called the Langmuir adsorption isotherm.
The Langmuir adsorption isotherm assumes that the gas attaches to the surface of the coal and covers the surface as a single layer of gas or monolayer. The Langmuir equation (or model) which describes the amount of gas stored on the surface of coal matrix as a function of pressure
If P = Pi, the relationship reflects initial gas content:
Nomenclature
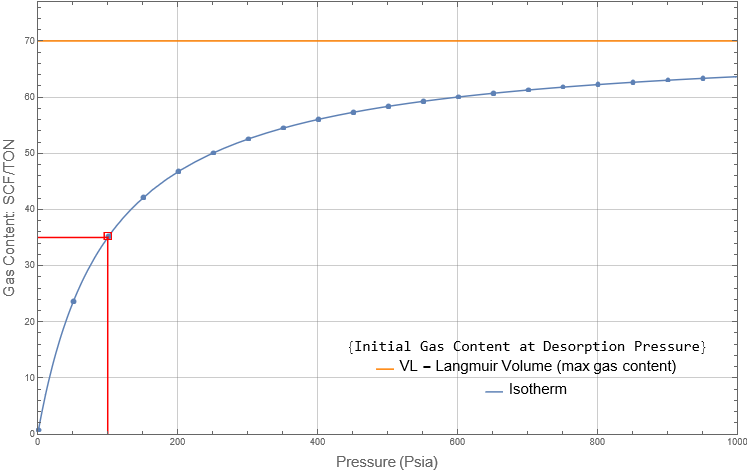
VL = Langmuir Volume: the maximum amount of methane absorbed on the surface of the coal matrix when the pressure, p, reaches infinity. This value is asymptotically approached by the isotherm as the pressure increases. The following figure is of a typical isotherm and shows its relationship with VL . In this example, initial reservoir pressure and desorption pressure are equal.
PL = Langmuir Pressure: the pressure where the amount of absorbed methane is 1/2 of the VL (which is the maximum amount of absorbed gas)
P = pressure of interest.
In some scenarios, we have the following:
b = 1/VL
According to Clarkson [2021], the popular Langmuir model is the least rigorous. There are an increasing number of improved models which can be found throughout the literature. These other models include:
Models other than the Langmuir model include:
-
Henry’s (Linear) adsorption isotherm (early 19th century)
-
Freundlick adsorption isotherm, 1909 - purely empirical
-
Brunauer-Emmett-Teller (BET) theory, 1938, JACS
Ultimately, natural gas in unconventional reservoirs consists of multiple hydrocarbons and nonhydrocarbon gas components. In coals, different components will absorb differently onto the internal surface area, as per Adsorption/ Desorption Mechanics
Gas Saturation
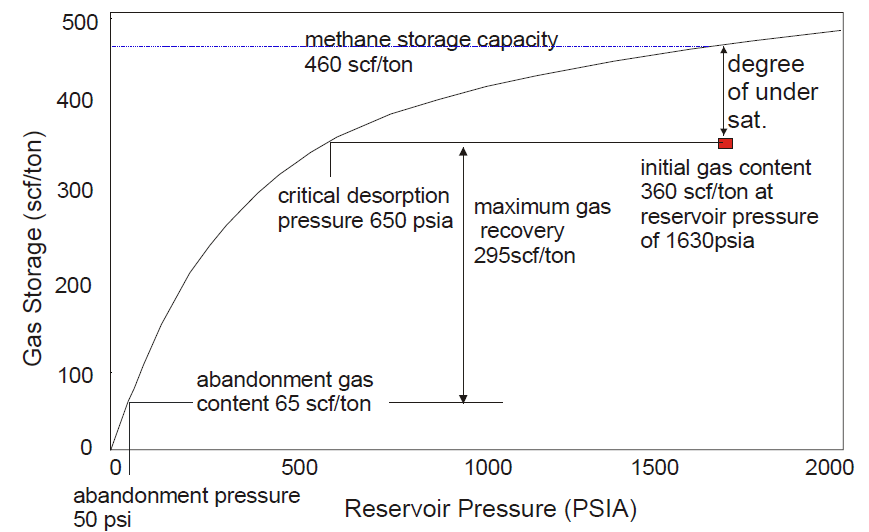
The degree of saturation of coal is expressed as a ratio of the adsorbed gas content to the adsorption capacity of the coal. The degree of saturation is important because:
-
the lower the saturation the less gas-in-place; and
-
The lower the saturation the greater the reservoir pressure decline required to produce adsorbed gas (I.e., significant dewatering)
See Also:
References
-
Zulkarnain, Ismail, Simulation study of the effect of well spacing, effect of permeability anisotropy, and effect of Palmer and Mansoori model on coalbed methane production, 2006 Texas A&M University, Texas A&M University,
-
Sinurat, Pahala Dominicus The Optimization of Well Spacing in a Coalbed Methane Reservoir, 2010, Texas A&M, Texas A&M
-
Bustin, R. M. ,Geology & Some Engineering Aspects of Coalbed Methane , 2001, CBM Solutions.
-
Clarkson, C. R. Unconventional Reservoir Rate-Transient Analysis: Volume I and II, 2021, Gulf Professional Publishing.
-
Thakur, Pramod, Advanced Reservoir and Production Engineering for Coalbed Methane
2017, Gulf Professional Publishing -
Zhang, Pengfei, Adsorption and Desorption Isotherms, Sept 3, 2016, KE Research Group