Introduction
The gas formation volume factor is used to relate the volume of gas, as measured at reservoir conditions, to the volume of the gas as measured at standard conditions, that is, 60°F and 14.7 psia. This gas property is then defined as the actual volume occupied by a certain amount of gas at a specified pressure and temperature, divided by the volume occupied by the same amount of gas at standard conditions. In equation form, the relationship is expressed as:
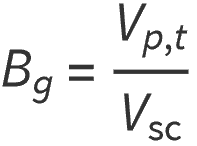
Where:
Bg = gas formation volume factor, ft3/scf
VP,T = volume of gas at pressure P and temperature T, ft3
VS = volume of gas at standard conditions
Applying the real gas equation , and subsituting for Volume V gives:
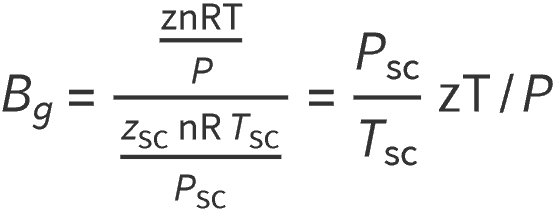
Where:
zSC = Z-factor at standard conditions (or 1.0)
PSC = standard pressure, often assumed to be 14.7
TSC = standard temperature, degree 520 oR (or 60 oF)
Subsituting the values for standard conditions gives:
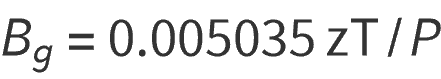
Bg = gas formation volume factor ft3/scf
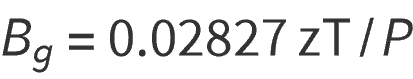
Bg = gas formation volume factor bbl/scf
A plot of Gas Bg is shown below using the Dranchuk, Purivs, and Robinson z-correlation at a gas gravity of 0.65, and a reference temperature of 120oF.
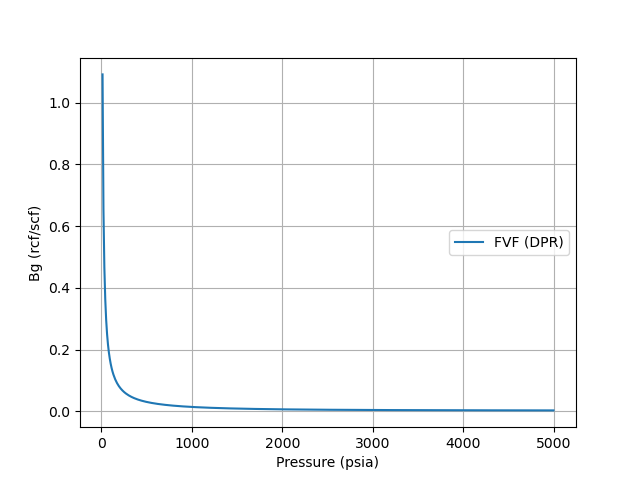
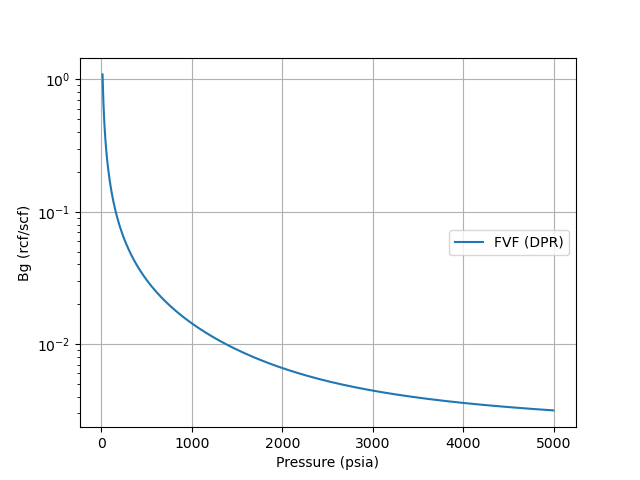
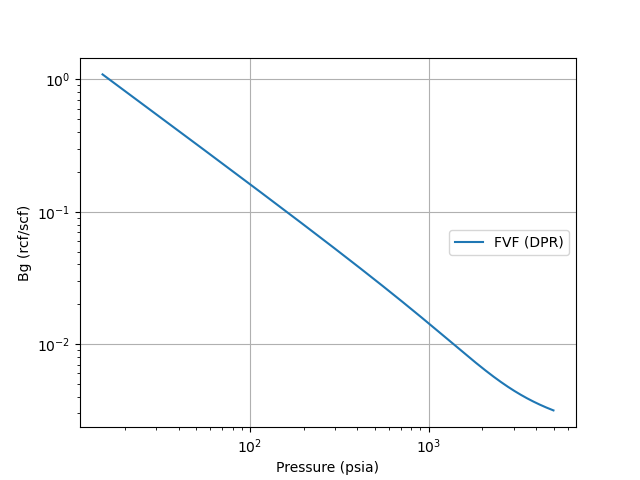
References
-
Ahmed, Tarek, Equations of Stateand PVT Analysis: Applications for Improved Reservoir Modelling , 2007, Gulf Publishing.
-
McCain, Willliam D., The Propertise of Petroleum Fluids, 1990, PennWell Books