Introduction
Also called the gas compressibility factor, z-factor, or supercompressibility, the gas deviation factor is defined as the ratio between the actual (or real) volume of gas and the ideal volume of gas. It is given by the following relationship:
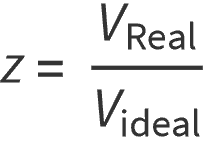
In other words:
Ideal Gas:
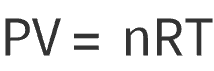
Real Gas:
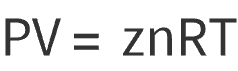
Where:
p = absolute pressure, psia
V = volume, ft3
T = absolute temperature, o
n = number of moles, lb-mole
R = Universal gas constant (10.73 psia ft3/lb-mole oR)
z = dimensionless z-factor (or gas compressibility factor)
Gas deviation factor is a measure of how the gas behavior deviates from the ideal behavior (given by the ideal gas law). It is determined in the laboratory by comparing the volume occupied by the gas under pressure and temperature with the volume of the gas calculated from the ideal gas law under the same pressure and temperature.
Gas deviation factor has been correlated with parameters that are readily available so it can be computed in absence of laboratory data. It approaches unity at pressures and temperatures close to standard pressure and temperature. The gas deviation factor is a property essential to determine for any further calculations involving dry gases.
The z-factor varies with any change in gas composition (i.e., molecular weight), as well as temperature and pressure. As described by some sources, at very low (near zero) pressures, the gas molecules are relatively far apart, which make gas behave as an ideal gas (the z-factor approaches 1.). By increasing pressure moderately, the molecules of gas become close enough to have some attaction among them resulting in less than ideal beahvoir. At higher pressures, the gas molecules are so close together that repulsive forces begin to apply and real gas volume approaches ideal gas and even results in z-factors greater than 1.0
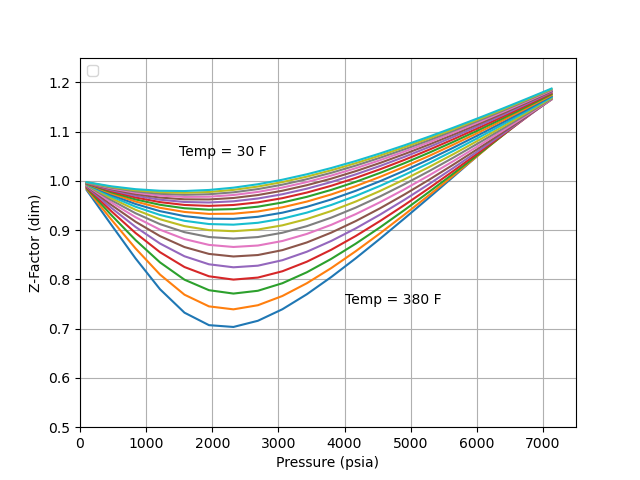
Standing and Kath presented a graphical generalized relationship of Z-factor, which is often considered an industry standard for predicting the volumetric beahvoir of natural gases. Many empirical equations and and EOS have been fitted to the Standing-Katz relationship.
The figure below shows a screen capture for z-factor at a 230 oF, and an gas gravity of 0.82. The plots show the correlations:
-
Beggs and Brill (1978)
-
Dranchuk and Abou-Kassam (1975)
-
Dranchuk et al (1974)
-
Papay (1985)
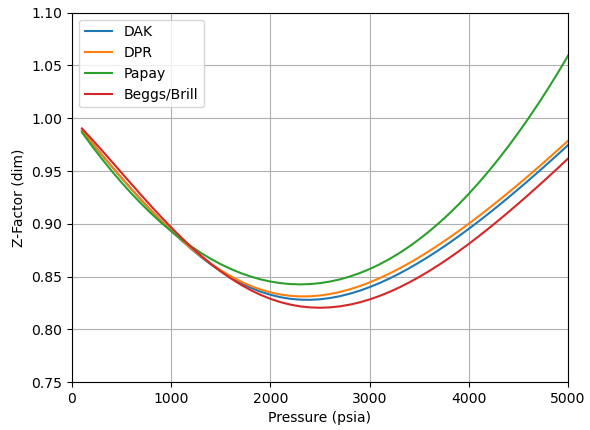
Gas deviation factor (z-factor) is used in the calculation of almost all PVT properties of gas. There are more than 20 known (and different) correlations for calculating z-factor. All of them compute z-factor from known pseudoreduced pressure and pseudoreduced temperature.
Dranchuk et al (1974)
Dranchuk, Purvis, and Robinson (1974) developed a correlation based on the Benedict- Webb-Rubin type of equation of state. Fitting the equation to 1500 data points from the Standing and Katz Z-factor chart optimized the eight coefficients of the proposed equations. The equation has the following form:
where:
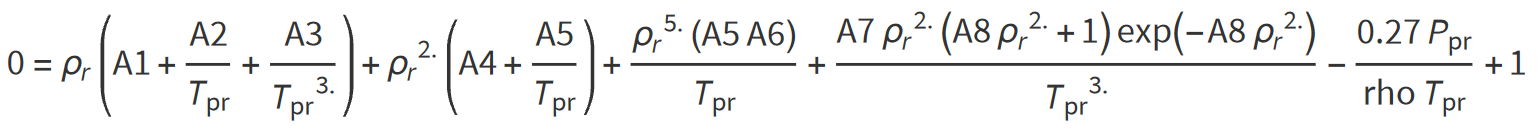
A1 = 0.31506237;
A2 = -1.0467099;
A3 = -0.57832729;
A4 = 0.53530771;
A5 = -0.61232032;
A6 = -0.10488813;
A7 = 0.68157001;
A8 = 0.68446549;
and
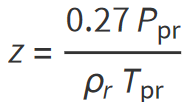
Dranchuk and Abou-Kassam (1975)
Dranchuk and Abu-Kassem (1975) derived an analytical expression for calculating the reduced gas density that can be used to estimate the gas compressibility factor. The reduced gas density ρr is defined as the ratio of the gas density at a specified pressure and temperature to that of the gas at its critical pressure or temperature:
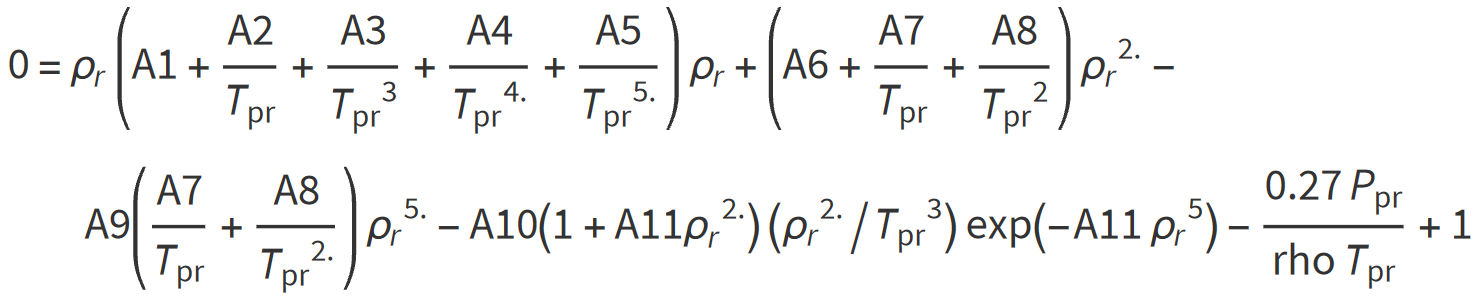
where:
A1 =0.3265
A2 =-1.0700
A3 =-0.5339
A4 =0.01569
A5 =-0.0516
A6 =0.5475
A7 =-0.7361
A8 =0.1844
A9 =0.1056
A10 =0.6134
A11 =0.7210
Papay
Papay (1985) proposed a simple expression for calculating the gas compressibility factor explicitly
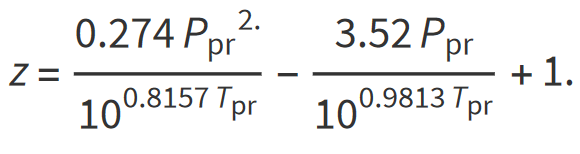
The is often considered an inferior approach.
Beggs & Brill (1978)
Another direct approach is that of Beggs and Brill (1978) as shown below:
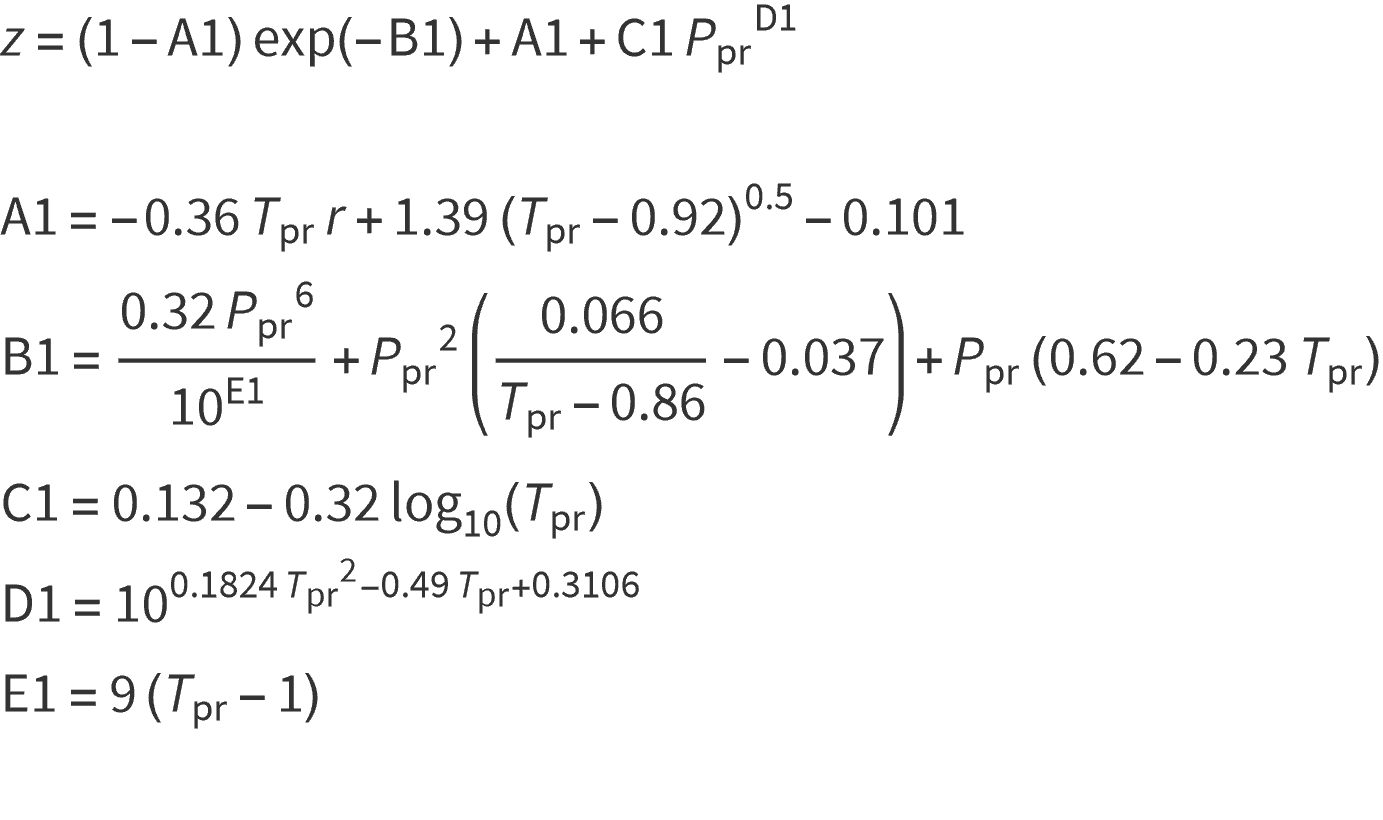
Computational Considerations
The equation which require an iterative solution can be solved for the reduced gas density ρr by applying the Newton-Raphson iteration technique as summarized in the following steps:
Step 1: Make an initial guess of teh uknown parameter, reduced gas density (ρr), as shown below:
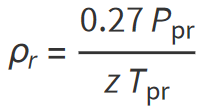
Step 2: Substitute the inital value for ρr into one the non-linear functions for z. Unless the correct value for ρr has been chosen, the non-linear z-function will have a non-zero value.
Step 3: A new and improved estimate of ρr can then be calculated following:
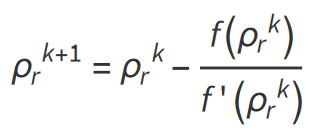
The steps are repeated until convergence according to a suitable criteria.
Finally
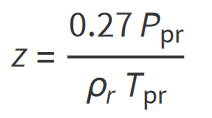
In the case of Dranchuk et al (1974):
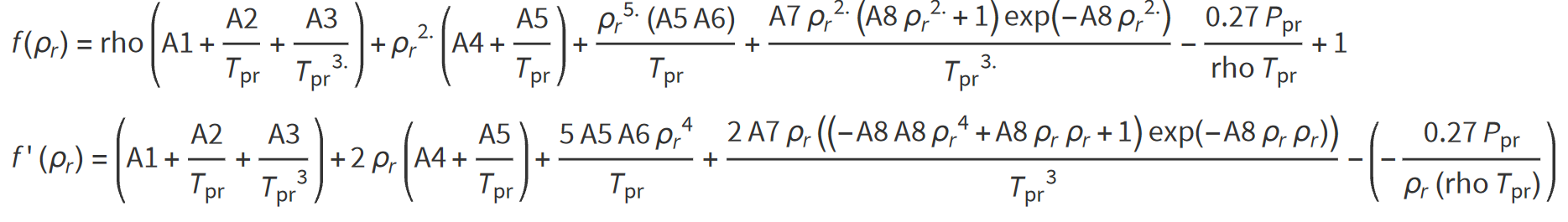
See Also:
References
-
El-Banbi, Ahmed / Alzahabi, Ahmed / El-Maraghi, Ahmed PVT Property Correlations: Selection and Estimation, 2018, Gulf Professional Publishing.
-
Ahmed, Tarek, Equations of State and PVT Analysis: Applications for Improved Reservoir Modelling, 2007, Gulf Publishing