Introduction
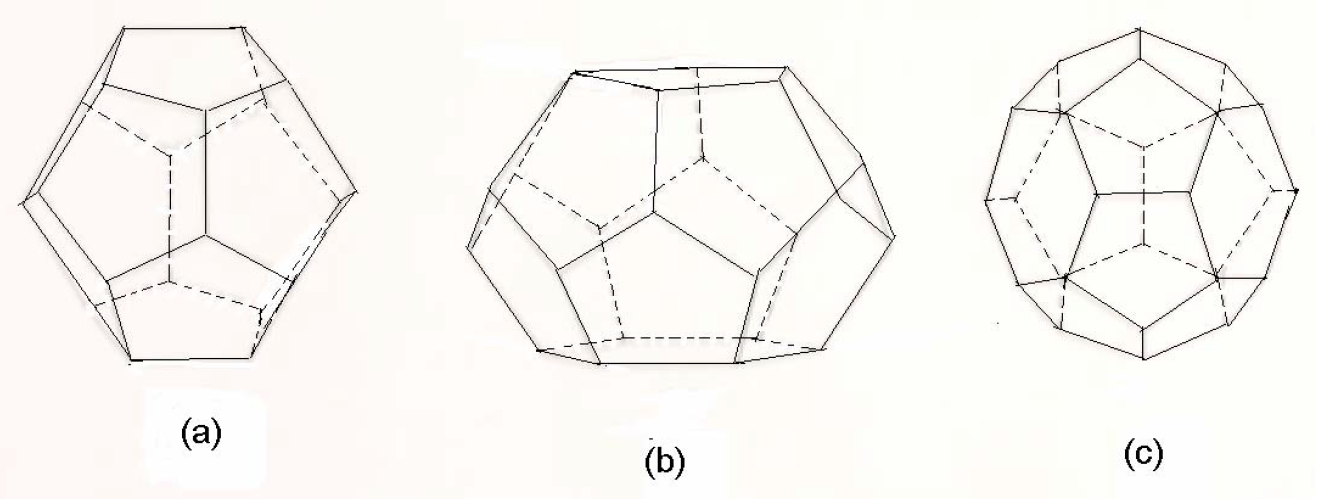
Gas hydrates are ice-like crystalline structures with gas components such as methane and carbon dioxide as guest molecules entrapped into cavities formed by water molecules.
Whenever a system of natural gas and water exists, hydrates can form especially at high pressure and low temperature.
In the oil and gas industry, gas hydrates are a serious problem in production and gas-transmission
pipelines because they plug pipelines and process equipment. By applying heat, insulating the pipelines, and using chemical additives as inhibitors, we can keep the operating conditions out of the hydrate-formation region.
Gas hydrates are nonstoichiometric compounds formed from mixtures of water and gas molecules under suitable pressures and temperatures. Gas molecules with adequate size become guest molecules entrapped in the cavities of cages formed by water molecules acting as host molecules. Hydrates are also called clathrates, which in Latin means, “cage.” When a minimum number of cavities are occupied by the gas molecules, the crystalline structure stabilizes and solid gas hydrates may form at temperatures above the
water freezing point. Most light molecules such as methane, ethane, propane, isobutane, normal butane, nitrogen, carbon dioxide, and hydrogen sulfide will form hydrates under specific conditions of pressure and temperature
References:
-
Ameripour, Sharareh (2005). Prediction of gas-hydrate formation conditions in production and surface facilities. Master's thesis, Texas A&M University. Texas A&M University