Introduction
Adsorption is a process by which certain porous solids bind large number of molecules to their surface. The number of molecules adsorbed on the solid surface depends on the pressure in the gas phase.
Tang et al. (2005) conducted gas sorption and displacement measurements on a cylindrical ground coal pack at room temperature (22 oC). Adsorption and desorption of pure CO2, N2, and CH4 was measured based on a gravimetric method at first escalating and then decreasing pore pressures. Experimental results are plotted in the figure below. Observations were:
-
The amount of adsorption (gas stored) increased with the increase of pressure.
-
At the same pressure, the amount of adsorption was the greatest for CO2, followed by CH4 and N2.
-
Methane and nitrogen are weakly absorbed species and are released before strongly adsorbed species such as CO2 and ethane.
-
According to Clarkson (2021), the amount of hydrocarbon component adsorption increases with molecular weight.
-
-
When decreasing the pressure, the desorption curve may not follow the same path down the adsorption curve (irreversible). At some pressure range, the desorption curve is above the adsorption curve, which forms a special shape in the isotherm plot.
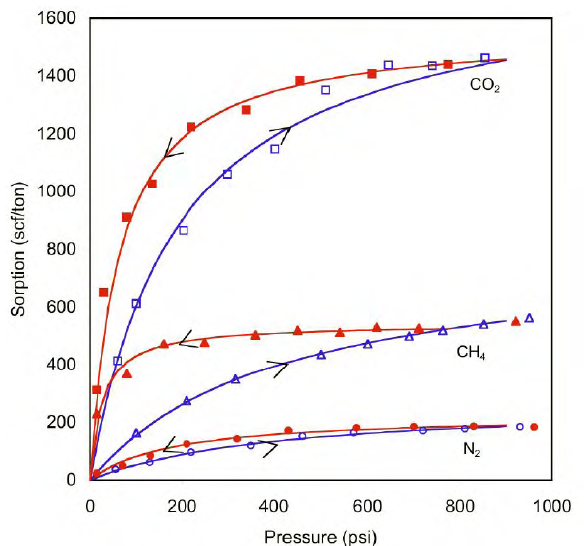
Given that the affinity of CO2 can be 2 to 3x that of methane, it can make it suitable candidate for ECBM and/or CO2 storage. Therefore, it is feasible to improve shale gas recovery by injecting CO2. Incidentally, according to Li et al (2020), CO2 does not easily desorbed in the production process.
Coals are also currently being investigated for sequestration of CO2 and H2S.
.
Incidentally, According to Hycal Energy Research Laboratories (2004), the affinity of H2S for CBM is even greater than that of CO2.
Practical Production Considerations
Given the varying infinities of methane and CO2 for coal, the produced gas stream from a well, project, or basin is characterized by steadily rising fractions of CO2 and heavier hydrocarbon gases. CO2 control becomes an increasing concern with CSG production.
See Also:
References
-
Lin, Wenjuan, Gas sorption and the consequent volumetric and permeability change of coal
2010, University of Standford,. -
Tang G.Q., Jessen K., and Kovscek A.R. 2005. Laboratory and Simulation Investigation of Enhanced Coalbed Methane Recovery by Gas Injection. Paper SPE 95947 presented at SPE Annual Technical Conference and Exhibition held at Dallas, Texas, U.S.A. on 8-12 October.
-
Tony Ma, Special Session 31: Back to Basics Series: An Introduction to Coalbed Methane, 2004 Canadian International Petroleum Conference.
-
Ying Sun Shuxia Li, Renyuan Sun Xiaoqiang Liu Hui Pu / Zhao, Jin, Study of CO2 Enhancing Shale Gas Recovery Based on Competitive Adsorption Theory, 2020 , ACS Omega
-
Clarkson, C. R., Unconventional Reservoir Rate-Transient Analysis: Volume I and II, 2021, Gulf Professional Publishing
-
John Seidle, Fundamentals of Coalbed Methane: Reservoir Engineering, 2011, PennWell Corporation.