Moisture
The graphic below shows that moisture has a strong effect on the adsorption capacity of the sample. The effect of H2O on the adsorption of CH4 is very obvious. The gas adsorption amount decreases when the content of water increases, and the difference rises with pressure increase. H2O is a polar molecule, and it is very easy to be adsorbed on the surface of minerals. Also, the diameter of H2O is smaller than that of CH4, which means H2O is more accessible to micropores. Therefore, the adsorption amount of CH4 decreases when moisture increases.
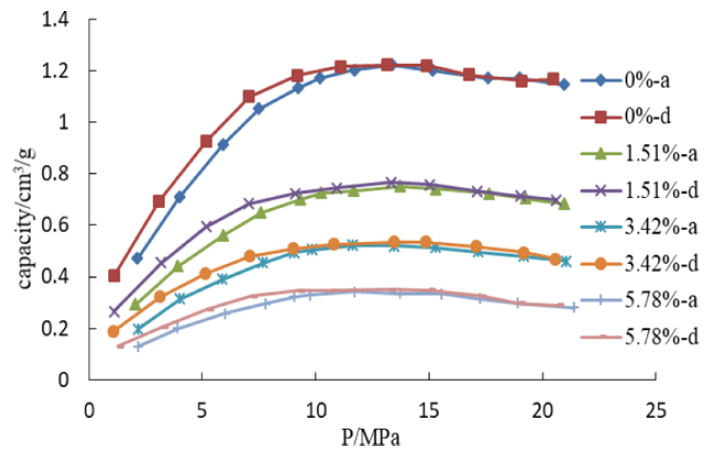
NOTE: In the above graphic “a“ represents adsorption isotherm, while “d“ represents desorption isotherm. There can be hysterias effects.
Ash
The ash content (the weight % residue after controlled combustion) of the coal is important in that is adsorbs little or no gas and hence dilutes the adsorption capacity of the whole coal.
See Also:
References
-
Ying Sun Shuxia Li, Renyuan Sun Xiaoqiang Liu Hui Pu / Zhao, Jin, Study of CO2 Enhancing Shale Gas Recovery Based on Competitive Adsorption Theory, 2020 , ACS Omega
-
Bustin, M., 2001, Geology and Some Engineering Aspects of Coalbed Methane, UBC.